Evaporative Air Coolers

What is an evaporative air cooler?
An evaporative air cooler, also known as a swamp cooler, is a cooling device that uses water to lower the temperature of the air in a room. There are some very active molecules in liquids flying out of the liquid surface and becoming vapor molecules when the air is unsaturated, this is evaporation.
Why does Evaporation Causes Cooling?
The molecules must have heat taken while flying out of the liquid into vapor. This heat is taken from the water that remains in the liquid state when evaporation occurs. It results in cooler liquid water. Evaporative cooling is a universal natural phenomenon. For example, in summer, we would feel cool when our wet-arm exposes to the air. It is because of the evaporation of water, which absorbs heat to cool our body, but not temperature shift. We feel cool if we sweat or sponge our bodies with alcohol. It is also because of evaporative cooling. In fact, evaporative cooling is a powerful and economical cooling technology.
There are four main factors affecting evaporation:
- Firstly, the relative humidity. There are always some molecules that fly out of the water into vapor molecules as long as the relative humidity of air is lower than 100% saturation. The lower the relative humidity, the faster the evaporation.
- Secondly, the temperature of the water, The higher the temperature of the water, the faster the water will be, and the more easily it will be evaporated into the air.
- Thirdly, the air velocity. The vapor molecules may return liquid water because of the collision with other vapor molecules. If the air flows fast, the vapor molecules have less chance to return, resulting in more evaporation
- Fourthly, the surface area. The molecular number near to air increases when the liquid surface area enlarges, more molecules may fly out of the liquid. Consequently, liquid surface area increases, evaporating faster.
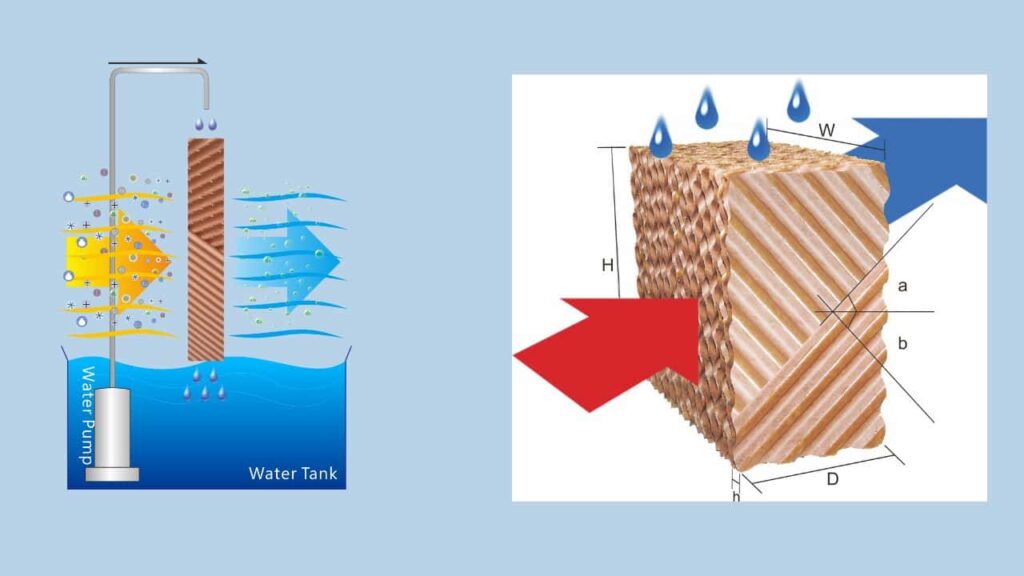
How does an evaporative air cooler work?
An evaporative air cooler works by drawing in hot air through a moistened pad or filter. The air passes through the pad, and as it does so, water evaporates from the pad, which cools the air.
Evaporative air coolers work by drawing hot, dry air into the unit and passing it over a wet pad. As the air passes over the wet pad, the water evaporates and cools the air. The cooled air is then circulated into the room, lowering the temperature and increasing the humidity level. The cooled air continues to circulate until the desired temperature and humidity level are reached, at which point the unit will automatically turn off.
Disadvantages of Evaporative Air Coolers
Limited effectiveness in high humidity
Evaporative air coolers are less effective in high-humidity environments, as the process of evaporation is less effective in these conditions. This makes them less suitable for use in areas with high levels of humidity, such as coastal regions.
Maintenance requirements
Evaporative air coolers require regular maintenance to ensure they continue to function effectively. This may include cleaning the pads and filters, topping up the water levels, and checking the fan and motor. Failing to carry out regular maintenance can lead to reduced efficiency and increased energy bills.
Limited cooling range
Evaporative air coolers are designed to cool the air by a few degrees, making them less suitable for use in extreme temperatures. Additionally, they may not be able to cool large spaces effectively, as the cool air may not be able to circulate properly.
Cost-effective
Evaporative air coolers are a much more cost-effective option compared to traditional air conditioning systems. They use significantly less energy to cool the air, making them an ideal choice for those looking to reduce their energy bills. Additionally, they are typically much less expensive to install and maintain, making them a more affordable option for those on a tight budget.
Eco-friendly
Evaporative air coolers are an environmentally-friendly option for cooling your home or office. They use a natural process to cool the air, which is significantly less harmful to the environment than traditional air conditioning systems that use refrigerants. Additionally, they use significantly less energy to cool the air, reducing your carbon footprint.
Improved air quality
Evaporative air coolers can help improve the air quality in your home or office. By increasing the humidity level in the air, they help to reduce the amount of dust and other pollutants in the air, making it cleaner and fresher to breathe. Additionally, they can help to reduce the risk of respiratory problems such as allergies and asthma.
Easy to install
Evaporative air coolers are relatively easy to install and do not require the services of a professional. They can be installed in a variety of different locations, making them an ideal choice for those with limited space or for those who do not have access to traditional air conditioning systems
Versatile
Evaporative air coolers are a versatile option for cooling your home or office. They can be used in a variety of different locations, making them an ideal choice for those who need to cool multiple rooms or large open spaces. Additionally, they come in a variety of different sizes, making them suitable for use in both small and large spaces.
